In Dusseldorf, Germany, global medical innovation forces gather together! From November 17th to 20th, 2025, the International Hospital and Medical Equipment and Supplies Exhibition (MEDICA2025) with the theme of “Meet Health.Future.People.” grandly opened at the local exhibition center. As a boundless communication platform that links global medical innovation and promotes the advancement of the health industry towards precision, efficiency and inclusiveness, this exhibition brings together top medical enterprises and core forces from home and abroad to jointly explore new opportunities for the development of the industry. Wuhan Life Origin Biotech Joint Stock Co.,Ltd., an innovative enterprise in China’s in vitro diagnostics field, made a grand appearance with a variety of core medical testing products and solutions, showcasing its “Made in China” diagnostic capabilities that are compliant and reliable and meet diverse clinical needs to global merchants.
In Dusseldorf, Germany, global medical innovation forces gather together! From November 17th to 20th, 2025, the International Hospital and Medical Equipment and Supplies Exhibition (MEDICA2025) with the theme of “Meet Health.Future.People.” grandly opened at the local exhibition center. As a boundless communication platform that links global medical innovation and promotes the advancement of the health industry towards precision, efficiency and inclusiveness, this exhibition brings together top medical enterprises and core forces from home and abroad to jointly explore new opportunities for the development of the industry. Wuhan Life Origin Biotech Joint Stock Co.,Ltd., an innovative enterprise in China’s in vitro diagnostics field, made a grand appearance with a variety of core medical testing products and solutions, showcasing its “Made in China” diagnostic capabilities that are compliant and reliable and meet diverse clinical needs to global merchants.
 The dry fluorescence immunoassay series equipment developed by Wuhan Life Origin Biotech creates a one-stop POCT solution. The sample types include serum, plasma, whole blood and peripheral blood of fingers, and multi-item quantitative analysis can be completed within 15 minutes. The equipment is easy to operate and does not require professional training. It is suitable for various scenarios such as home self-examination, hospitals, clinics, and health centers. It takes into account both the reliability of results and the convenience of operation, providing efficient support for clinical diagnosis.
The dry fluorescence immunoassay series equipment developed by Wuhan Life Origin Biotech creates a one-stop POCT solution. The sample types include serum, plasma, whole blood and peripheral blood of fingers, and multi-item quantitative analysis can be completed within 15 minutes. The equipment is easy to operate and does not require professional training. It is suitable for various scenarios such as home self-examination, hospitals, clinics, and health centers. It takes into account both the reliability of results and the convenience of operation, providing efficient support for clinical diagnosis.
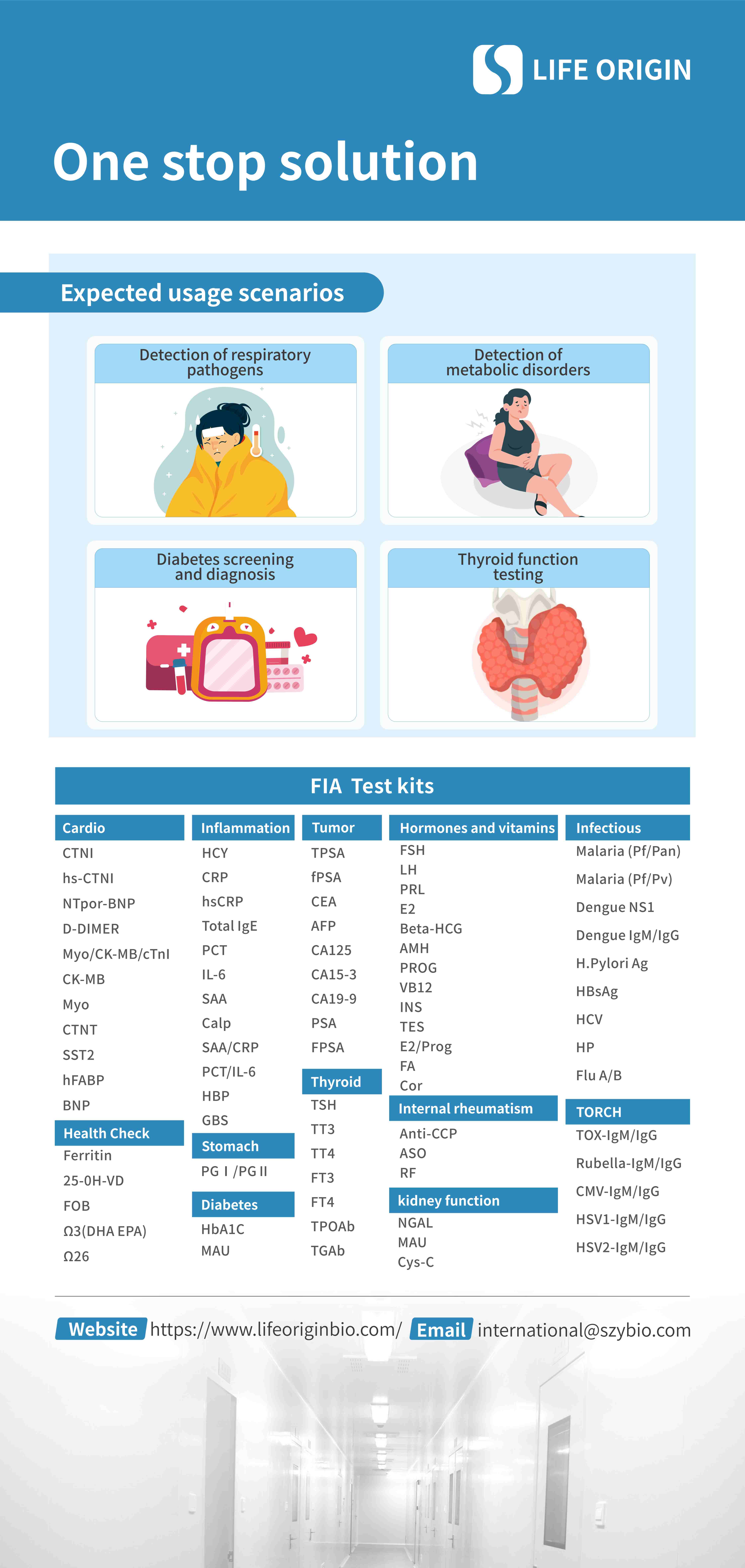
 POCT diagnostic reagent series and one-stop solutions, optimized and developed for the testing needs of different regions, are compatible with multiple mainstream testing platforms. This solution has a wide range of application scenarios and can cover diverse clinical needs such as respiratory pathogen detection, metabolic disorder detection, diabetes screening and diagnosis, and thyroid function testing. The FIA test kits that come with it cover a wide range of testing items in numerous fields such as heart, inflammation, tumors, vitamins, infectious diseases, thyroid, rheumatology and immunology, and renal function. The product strictly adheres to the requirements of the EU CE certification and the ISO 13485:2016 quality management system, is compatible with the voltage standards and clinical operation procedures of various countries, and can meet the clinical application needs of different countries and regions around the world, building a bridge for international medical cooperation and exchange.
POCT diagnostic reagent series and one-stop solutions, optimized and developed for the testing needs of different regions, are compatible with multiple mainstream testing platforms. This solution has a wide range of application scenarios and can cover diverse clinical needs such as respiratory pathogen detection, metabolic disorder detection, diabetes screening and diagnosis, and thyroid function testing. The FIA test kits that come with it cover a wide range of testing items in numerous fields such as heart, inflammation, tumors, vitamins, infectious diseases, thyroid, rheumatology and immunology, and renal function. The product strictly adheres to the requirements of the EU CE certification and the ISO 13485:2016 quality management system, is compatible with the voltage standards and clinical operation procedures of various countries, and can meet the clinical application needs of different countries and regions around the world, building a bridge for international medical cooperation and exchange.
As a Chinese high-tech enterprise deeply engaged in the field of in vitro diagnostics (IVD), Wuhan Life Origin Biotech has always taken compliant R&D as the foundation and technological innovation as the core driving force. During the MEDICA2025 exhibition, the company made a grand appearance with its complete industrial chain layout (raw material research and development → reagent production → finished product sales), diversified testing solutions and integrated OEM/ODM services, deepening exchanges and cooperation with global medical industry partners. Relying on the support of international certification systems such as CE and ISO, the company promotes diagnostic products and services that meet clinical needs and international standards to the world, injecting Chinese strength into the global health cause. In the future, Wuhan Life Origin Biotech will continue to link global resources with an open attitude, join hands with industry colleagues to promote the innovation and upgrading of in vitro diagnostic technology and the popularization of its application, and contribute to the high-quality development of the global medical and health industry.
Post time: Nov-21-2025